Overall Survival: The Gold Standard Under Scrutiny

Overall survival (OS) has long been considered the gold standard in oncology trials. Defined as the time from treatment initiation until death from any cause, OS provides a direct and intuitive measure of a therapy's impact on patient longevity. However, as the costs of oncology treatments skyrocket, even OS is coming under scrutiny. Cases like erlotinib (Tarceva) in pancreatic cancer and bevacizumab (Avastin) in breast cancer highlight how small OS gains may fail to justify the high costs, both financially and in terms of side effects. Moreover, payers in Europe and the United States are increasingly questioning whether such marginal benefits provide meaningful value for patients.
What Is Overall Survival?
OS is calculated using Kaplan-Meier survival analysis. The formula for the survival probability at time ttt is:
$$
\text{OS}(t) = \prod_{t_i \leq t} \left( 1 - \frac{d_i}{n_i} \right)
$$
where:
- ti: Time of the iii-th death,
- di: Number of deaths at ti,
- ni: Number of patients at risk just before ti.
This approach generates a stepwise survival curve showing the proportion of patients alive over time.
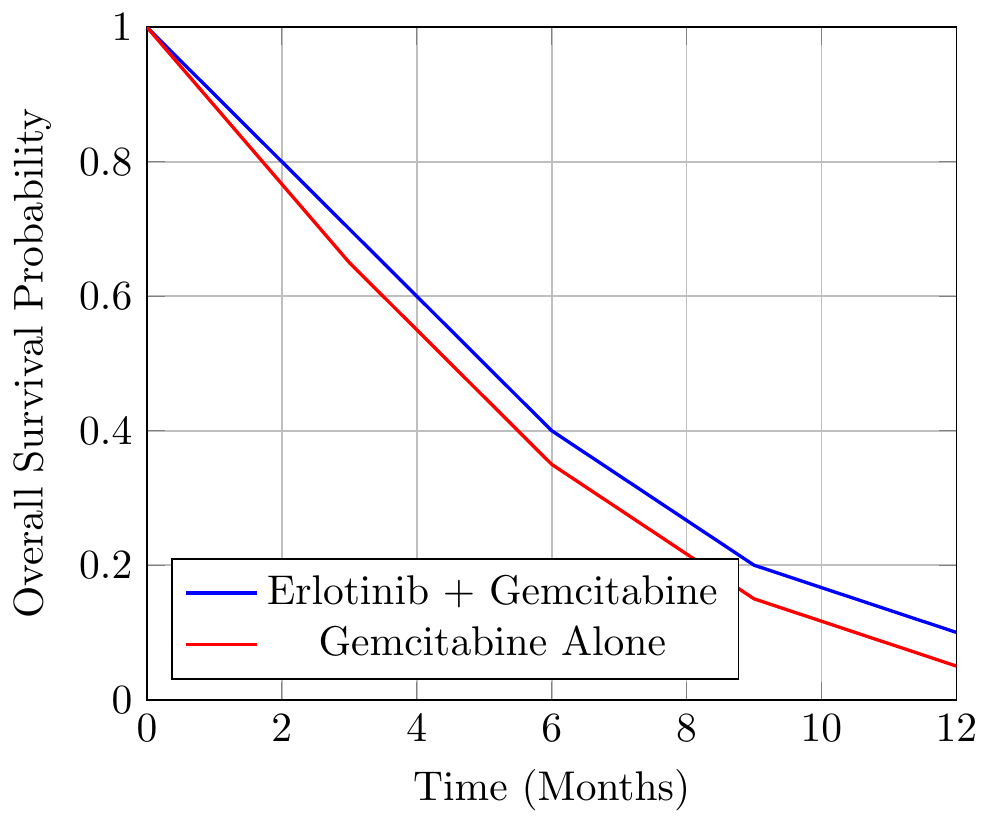
Case Study: Erlotinib in Pancreatic Cancer
ECase Study: Erlotinib in Pancreatic Cancer
Erlotinib, marketed as Tarceva, was approved in combination with gemcitabine for advanced pancreatic cancer based on a statistically significant improvement in overall survival (OS). However, the clinical relevance of the gain—just 0.5 months—was widely debated.
Trial Results
| Treatment Regimen | Median OS (Months) | Improvement |
|---|---|---|
| Gemcitabine Alone | 5.9 | – |
| Erlotinib + Gemcitabine | 6.4 | +0.5 |
Visualizing OS Data
This modest improvement can be illustrated with a Kaplan-Meier plot:
Cost Considerations
- Erlotinib Cost: 30 tablets (100 mg) cost approximately $4,553, or $151 per tablet.
- Standard Dosage: At 100 mg daily, the monthly cost for erlotinib is approximately $4,553.
- Gemcitabine Cost: A six-month course is approximately $23,500, or about $3,917 per month.
- Combined Monthly Cost: Erlotinib plus gemcitabine totals approximately $8,470 per month.
- Other Costs: These figures exclude administration fees, supportive care, and side effect management, which further increase the overall expense.
Criticism
- Marginal Benefit: Erlotinib added only 0.5 months (15 days) to median OS.
- No Quality of Life Improvement: Patients experienced side effects such as rash and diarrhea, with no reported quality of life benefits.
- High Cost: Combined treatment costs exceeded $8,000 per month, raising significant cost-effectiveness concerns.
European Case Study: Bevacizumab in Breast Cancer
Bevacizumab (Avastin) was initially approved in Europe for metastatic breast cancer based on progression-free survival (PFS) improvements. However, subsequent analyses revealed no OS or quality-of-life benefits, leading to its withdrawal in this indication.
PFS vs. OS
While bevacizumab improved PFS by several months, OS remained unchanged:
| Endpoint | Bevacizumab + Chemo | Chemo Alone | Improvement |
|---|---|---|---|
| Median PFS | 11.3 months | 6.7 months | +4.6 months |
| Median OS | 25.2 months | 24.1 months | +1.1 months |
Outcome
In 2012, the European Medicines Agency (EMA) withdrew its approval for bevacizumab in breast cancer, citing a lack of clinically meaningful OS benefit and concerns about toxicity.
Criticisms of OS as an Endpoint
- Marginal Gains:
- OS benefits are often small, as in erlotinib (0.5 months) or bevacizumab (1.1 months). These modest improvements may not justify high treatment costs.
- Cost vs. Value:
- For drugs with marginal OS benefits, incremental cost-effectiveness ratios (ICERs) often exceed thresholds acceptable to payers (e.g., €50,000–€100,000 per QALY in Europe).
- Confounding Factors:
- OS can be influenced by subsequent lines of therapy, making it difficult to attribute gains solely to the drug being studied.
- Impact on Healthcare Budgets:
- Funding therapies with marginal OS benefits diverts resources from treatments that provide greater value, such as immunotherapies with durable responses.
Conclusion
Overall survival remains the cornerstone of oncology trials, but its role is increasingly questioned when marginal improvements are paired with high costs and toxicity. The cases of erlotinib and bevacizumab highlight the tension between statistical significance and clinical relevance. For payers in Europe and elsewhere, these examples underscore the need for therapies to demonstrate not just longer lives, but better lives—at a cost society is willing to bear.
Member discussion